Living Colors And New Renewable Energy Sources: plant metabolites in solar cells
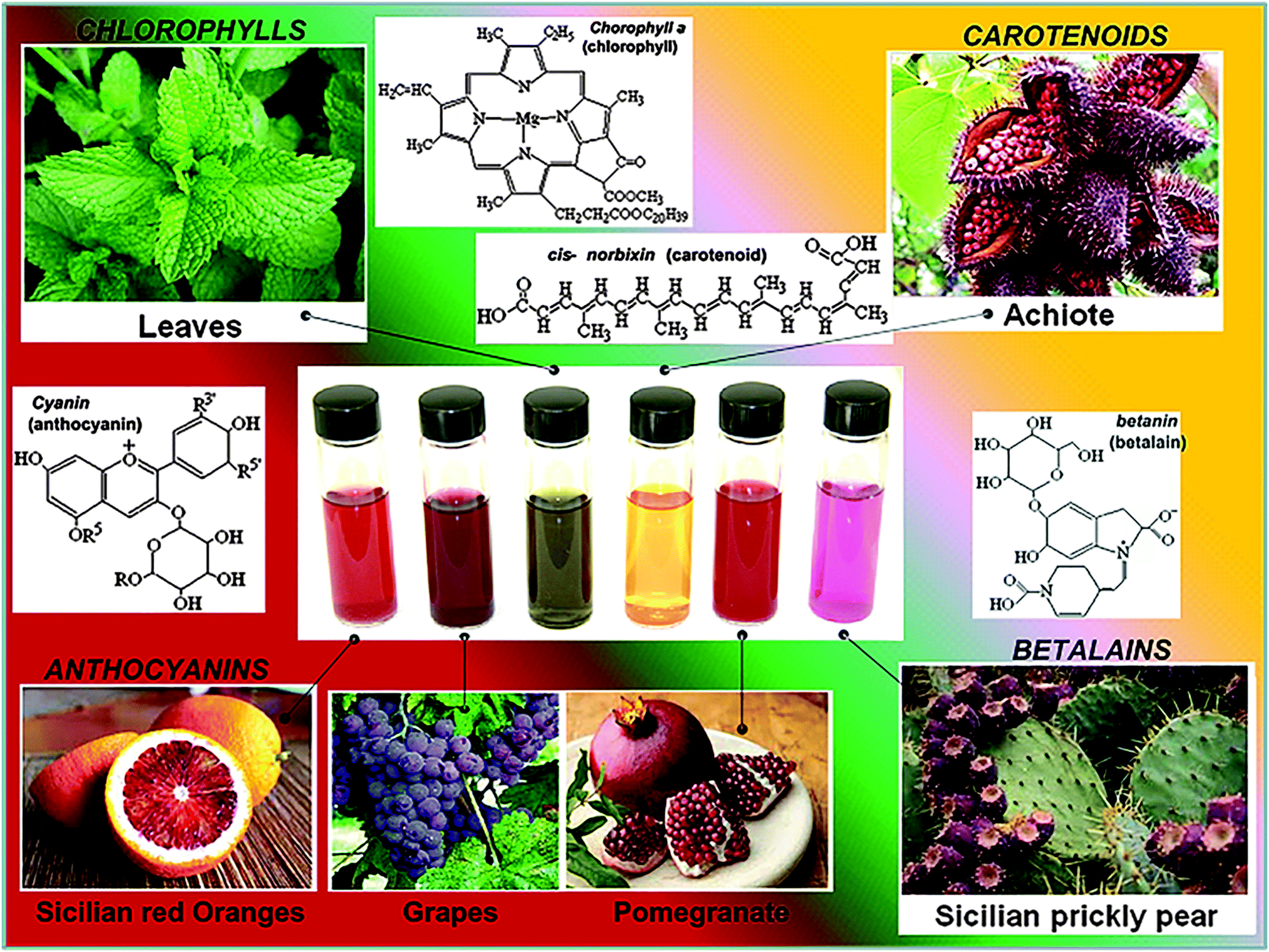
Many biological molecules give color to living objects by absorbing light of particular colors and reflecting the remaining light. One of the best known examples is chlorophyll: an extremely important biomolecule critical in photosynthesis that allows plants to absorb energy from light. Several types of chlorophyll and closely related porphyrin-based structures are found in green pigments in cyanobacteria and the chloroplasts of algae and plants. The function of the reaction center of chlorophyll is to use the energy absorbed by and transferred to it from the other groups in the photosystem to donate an electron into a series of molecular intermediates called an electron transport chain.
The other significant group of plant dyes consists of mixes of anthocyanins belonging to a parent class of flavonoids. In photosynthetic tissues (such as leaves and sometimes stems), anthocyanins have been shown to act as a "sunscreen", protecting cells from high-light damage, by absorbing blue-green and ultraviolet light and protecting the tissues from photoinhibition, or high-light stress.

In biophotovoltaic dye-sensitized solar cells the dye-sensitizer mimics nature in absorbing sunlight and transforming solar energy into electricity. A modern biomimetic solar cell usually consists of arrays of titania nanocrystals coated with light-sensitive natural or synthetic pigments playing the role equivalent to chlorophyll in nature. The many benefits of DSCs include lower purity requirements and abundance of component materials, as well as the fact that they can be produced on flexible substrates, making them amenable to roll-to-roll printing processes. Their advantages are based on low cost of production, ease of fabrication and modifiable aesthetic features, such as color and transparency [1]. DSCs are emerging as one of the most promising low cost photovoltaic technologies, addressing secure, clean, and efficient solar energy conversion.
Chlorophyll's and anthocyanin's abitily to convert light energy into electrical energy makes them prime candidates for use in DSC dye-sensitiers. In a natural solar cell the chlorophyll molecules absorb light most strongly in the red and blue parts of the spectrum, leaving the green light to be reflected. The absorbed energy is sufficient to free an electron from the excited chlorophyll. Chlorophyll, cyanin, and carotene are several of the natural dyes that have been succesfully utilized in DSCs. Others include phthalocyanine and porphyrin dyes developed based on natural prototypes. Numerous metal complexes and organic dyes have been synthesized, many of them mimic chlorophyll-like structures (for examples zinc-porphyrin-based light sensitizers).
Porphyrins are water-soluble biological pigments widely spread in animal and plant tissues. They take part in important biological functions such as metal-binding cofactors in animal hemoglobins, chlorophyll for photosynthesis, and certain enzymes for cell respiration. One of the best-known porphyrins is heme, the pigment in red blood cells, a cofactor of the protein hemoglobin. The specific porphyrin in heme B is called protoporphyrin IX and has 4 methyl, two vinyl, and two propionic acid substituents.
Numerous types of porphyrins can be obtained through the modification of a natural porphyrins or through chemical synthesis. Not only their chemical transformations through nucleophilic and electrophilic substitution, substituent modification, reduction, oxidation but also their complex formations with iron, zinc, copper, nickel and cobalt yield valuable biological applications in molecular biology, fluoroimmunoassay and new materials science.
Compounds similar to porphin are the parent compounds for similar ring systems with other central metal atoms in biochemistry. These include chlorin, which has a magnesium atom in several types of chlorophyll; bacteriochlorin, found in bacteriochlorophyll; and corrin, which has a cobalt atom in cobalamin or vitamin B12.
Another class of molecules with technological applications in biophotovoltaics is phenolic metabolites, e.g. natural flavonoids and xanthonoids. Twenty natural dyes extracted from flowers, leaves, fruits, and traditional Chinese medicinal herbs, have been successfully used as sensitizers in DSCs [2]. The list includes natural dyes extracted from begonia, tangerine peels, rhododendron, yellow rose, petunia, mangosteen pericarp, coffee and other common plant materials. A dye mix of particular interest obtained from mangosteen pericarp was found to contain xanthonoid mangostin and flavonoid-based rutin.
Rutin is a plant pigment that is found in several fruits and vegetables. Rutin is used to make medicine. The major sources of rutin for medical use include buckwheat, Japanese pagoda tree, and eucalyptus, lime tree flowers and elder flowers and hawthorn leaves. Anthocyanins and betalains from many plant sources were also established as promising light-harvesting substances for dye-sensitized solar cells [3].
In various scientific studies related to the subject, NMR spectroscopy is often used as a powerful tool in elucidating the structure of color-inducing metabolites, particularly to follow the structural changes in the natural compound mixes. In modern structural studies of natural dyes, a complete 1- and 2D 1H NMR and 13CNMR experiment set is a method of choice. The metabolomics database at the BMRB contains 1H, 13C, 13CDEPT90, DEPT135, TOCSY, COSY45, HSQC, HMBC and HSQC-TOCSY-ADIA NMR spectral data for various carotenoids, flavonoids and porphyrins, including such derivatives as zinc tetraphenylporphine, protoporphyrin IX, and rutin. They are presented as the time domain data, the spectra images, and the tables of assigned chemical shifts.
1.
"Dye-sensitized solar cells", Grätzel, M;
Journal of Photochemistry and Photobiology C: Photochemistry Rewiews 4 (2): 145-53, 2003
(doi:10.1016/S1389-5567(03)00026-1)
2.
"Dye-sensitized solar cells using 20 natural dyes as sensitizers", Huizhi Zhou et al; J. Photochem.Photobiol. A: Chemistry,
219(2-3), 188-194, 2011
(doi:10.1016/j.jphotochem.2011.02.008)
3.
"Vegetable-based dye-sensitized solar cells",
Calogero G et al.; Chem.Soc.Rev., 2015, 44, 3244 (DOI: 10.1039/C4CS00309H)