Flavonoids
Flavonoids (and bioflavonoids) are a class of plant secondary metabolites. The name is coming from the Latin word flavus meaning yellow, their color in nature.
Based on the differences in the structure of the aglycones carbon ring, flavonoids can be classified into seven groups: flavones, flavanones, flavonols, flavanonols, isoflavones, flavanols (catechins) and anthocyanidins.
Bioflavonoids, or flavonoids found in nature, are abundant in the pulp and rinds of citrus fruits and other foods containing vitamin C, including red wine, green tea, onions, grapefruit seeds, apples, and other fruits and berries. Many medicinal plants contain bioflavonoids, such as ginkgo biloba, hawthorn, and Chinese scullcap. Citrus flavonoids are found in citrus fruits, such as rutin in buckwheat, epigallocatechin gallate in green tea, anthocyanidins in bilberry, and naringenin in grapefruit. Oligomeric proanthocyanidins are found in grape seeds and skins. Quercetin is found in onions, tea, and apples (Fig.1).
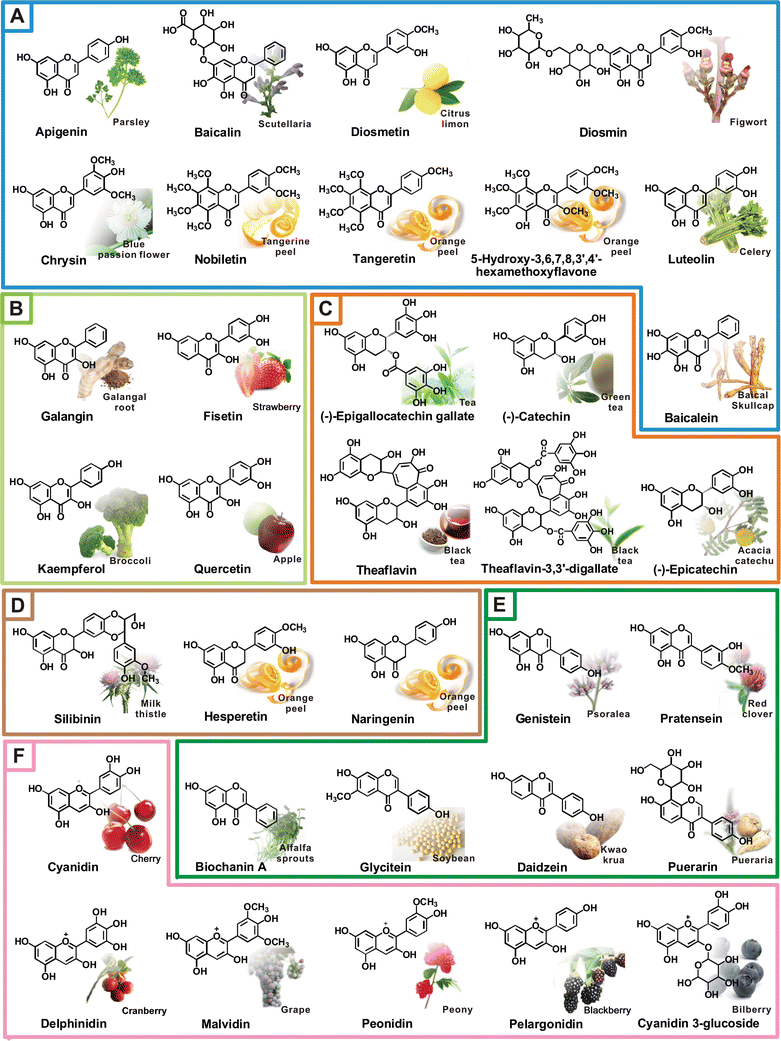
Fig.1 Bioflavonoids and their sources (from "Anti-inflammatory activity of natural dietary flavonoids"[1].)
 Fig. 2 Traditional Chinese herbs
Fig. 2 Traditional Chinese herbs
Structurally related isoflavones are found mainly in soy and products made from soy. They are derived from the flavonoid biosynthesis pathway via liquiritigenin or naringenin. While flavonoids (in the narrow sense) have the 2-phenylchromen-4-one backbone, isoflavonoids have the 3-phenylchromen-4-one backbone. Naringenin: BMRB entry bmse001121, a very significant metabolite, is a flavanone, a type of flavonoid, that is considered to have a bioactive effect on human health as an antioxidant, free radical scavenger, anti-inflammatory agent, carbohydrate metabolism promoter, and immune system modulator. It is the predominant flavanone in grapefruit.
Ever since ancient times, people looked for healing substances in nature. Flavonoids are widely present in herbs and plants, and have been demonstrated to exhibit a broad spectrum of biological activities for human health. Numerous studies have proposed that flavonoids act through a variety mechanisms to prevent and attenuate inflammatory responses and serve as possible cardioprotective, neuroprotective and chemopreventive agents.
One of such plants, Baikal Skullcap or Scute, is a herb from the lamiaceae (mint) family and is native to China. "Its Pinyin name is Huang Qin, it is very bitter and has a cold energetic nature" is written in ancient Chinese texts. In traditional Chinese Medicine (Fig. 2) this plant was used (almost always in combination with other plants or foods), to "clear heat, dry damp, drain fire, and resolve toxins", i.e. to support normal immune function. This plant is very chemically complex and is full of antioxidant flavones that appear to down regulate the effects of oxidative stress on various tissues in the body, including the liver. It also supports the body in having a healthy inflammatory response and promotes normal cellular growth.
In a world of plants and Rhizobia symbiosis, flavonoids are secreted by the root of their host plant. That helps Rhizobia in the infection stage of their symbiotic relationship to accommodate with legumes like peas, beans, clover, and soy.
On the other hand, the major rice pathogens: Xanthomonas oryzae pv. Oryzae (the bacterial blight pathogen), Pyricularia oryzae (the fungal blast pathogen), and Rhizoctonia solani (the fungal sheath blight pathogen) are fraught with several flavonoids with variable activity. Naringenin inhibited the growth of X. Oryzae, naringenin and kaempferol - the spore germination of P. oryzae.
In addition, flavonoids have been used to characterize wines. For example, in Pinot Noir wine, the highest concentrations of catechin and epicatechin were found, while Cabernet Sauvignon contains quercetin (typical of red wines).
Naringenin's apparent ability to lower cholesterol concentration and protect from obesity effects of a high-fat diet, and high concentrations of flavonoids in red wines bring us to an intriguing phenomenon of Mediterranean Diet, or French Paradox. The studies conclude that a diet based on Mediterranean cuisine which is high in omega-3 oils, antioxidants and includes moderate consumption of red wine, is partially responsible for lower incidence of cancer, myocardial infarction and cardiovascular disease, partly through increasing HDL cholesterol whilst reducing LDL cholesterol.
Several recent research articles have demonstrated the efficient production of flavonoid molecules from genetically engineered microorganisms. E. coli cells containing the gene clusters produced two flavanones, pinocembrin from phenylalanine and naringenin from tyrosine, in addition to their precursors: cinnamic acid and 4-coumaric acid. This is a new exiting metabolic engineering technique for the production in bacteria of a variety of compounds of plant and animal origin [2]. In different studies, metabolic engineering of the complete pathway led to biosynthesis of various flavonoids and stilbenoids in Saccharomyces cerevisiae [3].
Nuclear magnetic resonance (NMR) spectroscopy is increasingly used as a technique to provide insight into mixture of natural products belonging to the same or different chemical classes without previous separation of the individual components. The sample preparation for NMR is simple and nondestructive. In this context, NMR methods have been used with success in the structural identification of natural products such as flavonoids and derivatives in mixture. The metabolomics database at the BMRB contains 1H, 13C, 13CDEPT90, DEPT135, TOCSY, COSY45, HSQC, HMBC and HSQC-TOCSY-ADIA NMR spectral data for compounds related to flavonoids research (such as naringenin, quercetin, kaempferol, genistein etc) presented as time domain data. The spectra pictures as well as the tables of peak transitions and assigned chemical shifts are also available. Besides direct research, the data are of interest for building of the modular suites of exercises for laboratory sessions in Physical Chemistry and Instrumental Analysis, and specifically useful for teaching the 1D and 2D 1H-13C data assignments by providing visual context.
1. "Anti-inflammatory activity of natural dietary flavonoids" (Review Article), Min-Hsiung Pan, Ching-Shu Lai and Chi-Tang Ho; Food Funct. 2010, 1, 15-31 (doi:10.1039/C0FO00103A.)
2. "Production of plant-specific flavanones by Escherichia coli containing an artificial gene cluster", Hwang E I, Kaneko M, Ohnishi Y, Horinouchi S; Appl. Environ. Microbiol. 2003 May, 69 (5): 2699..706 (doi:10.1128/AEM.69.5.2699-2706.2003. PMC:154558. PMID:12732539.)
3. "Metabolic engineering of the complete pathway leading to heterologous biosynthesis of various flavonoids and stilbenoids in Saccharomyces cerevisiae", Trantas E, Panopoulos N, and Ververidis F; Metabolic Engineering 08/2009; 11 (6): 355..366. (doi:10.1016/j.ymben.2009.07.004. PMID:19631278.)
